
The sulfur-containing thiol group in cysteine’s side chain is key to its properties, enabling the formation of disulfide bridges between two peptide chains (as with insulin) or loop formation within a single chain, impacting the final protein structure. Subsequently, it was chemically synthesized, and the structure solved in 1903–4.
The water solubility of this amino acid lends itself to the presence near the active sites of enzymes such as pepsin.Ĭysteine is particularly abundant in the proteins of hair, hooves, and the keratin of the skin, having been isolated from a urinary calculus in 1810 and from horn in 1899. Asparagine is important in the removal of toxic ammonia from the body.ĭiscovered in proteins in 1868, aspartic acid is commonly found in animal proteins, however only the l-stereoisomer participates in the biosynthesis of proteins. Only the l-stereoisomer participates in the biosynthesis of mammalian proteins. However, it wasn’t until 1932 that scientists were able to prove that asparagine occurred in proteins. In 1806, asparagine was purified from asparagus juice, making it the first amino acid to be isolated from a natural source. Although arginine is necessary for growth but not body maintenance, research has indicated that arginine is crucial to the wound-healing process, particularly in those with poor circulation. Arginine can also be produced synthetically, and arginine-related compounds can be used in treating people with liver dysfunction due to its role in promoting liver regeneration. It can then be converted into nitric oxide by the human body, a chemical known to relax blood vessels.ĭue to its vasodilatory effects, arginine has been put forward for the treatment of people with chronic heart failure, high cholesterol, compromised circulation and high blood pressure, although research on these fronts is still ongoing. In humans, arginine is produced when proteins are digested. Only the l-stereoisomer participates in the biosynthesis of proteins. Its low reactivity contributes to the simple, elongated structure of silk with few cross-links which gives the fibers strength, stretch resistance and flexibility. Finally, abbreviations used for amino acid residues with more than one potential identity, and the termination codon are shown in red to complete the alphabet of single letter abbreviations.ĭiscovered in protein in 1875, alanine makes up 30% of the residues in silk. In addition, pyrrolysine, used in the biosynthesis of proteins in some archaea and bacteria but not present in humans, and selenocysteine, a cysteine analogue only found in some lineages, are included in blue. This table shows the abbreviations and single letter codes used for the 20 amino acids found in proteins.

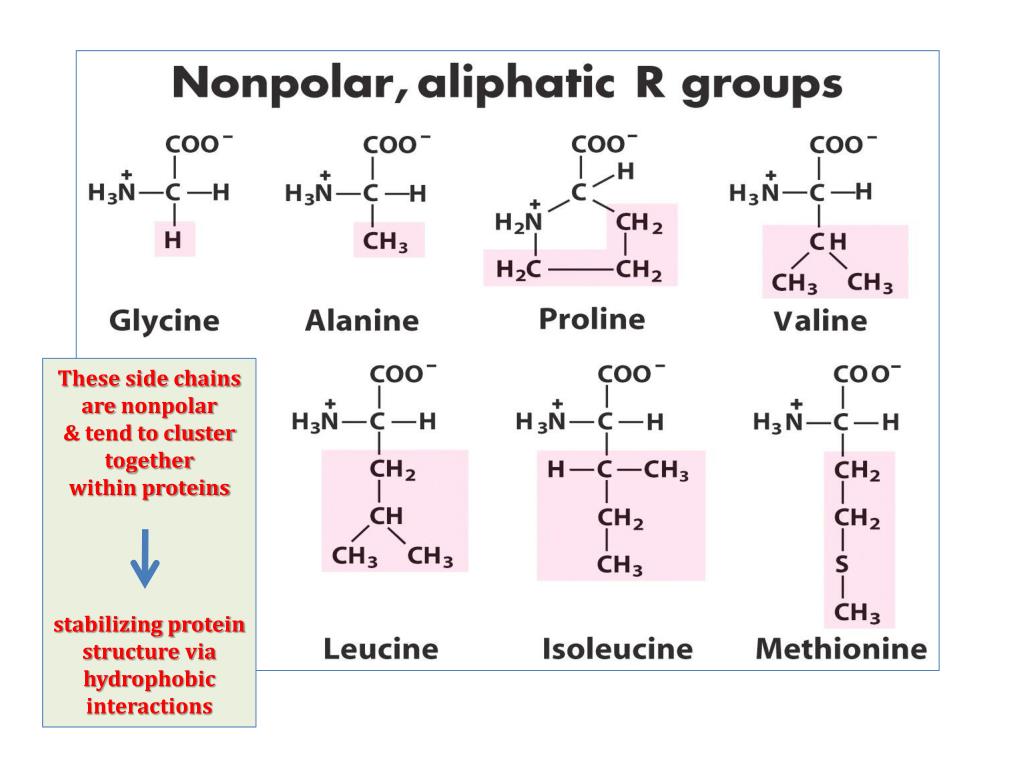
This chart shows the chemical structures of the 20 amino acids that make up proteins.
